Accurate chlorine testing is crucial for water quality management. The DR300 pocket colorimeter chlorine can provide reliable readings. This compact device is designed for easy handling and portability. It helps users analyze water samples with precision.
Understanding how to operate the DR300 pocket colorimeter chlorine is essential. If used incorrectly, results may vary significantly. Users must ensure proper calibration before testing. A well-prepared sample leads to more accurate outcomes. Even slight mistakes can alter the readings, impacting water safety.
In this guide, we will explore the steps to effectively utilize the DR300 pocket colorimeter chlorine. Each step is designed to enhance accuracy and reliability. This device can be an invaluable tool for professionals and enthusiasts alike. Yet, it requires careful handling and attention to detail. Let's dive into the process of achieving accurate chlorine tests.
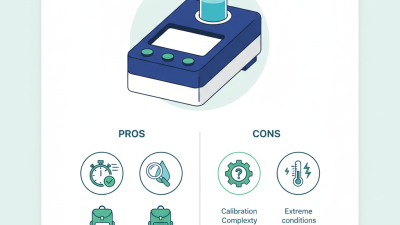
The DR300 Pocket Colorimeter is a handy tool for measuring chlorine levels in various water sources. It is compact and easy to use, making it accessible for many users. Understanding its basic features is crucial to achieving accurate results. The device typically includes a simple interface with a digital display, making readings clear and straight to the point. It's essential to ensure that each test is performed properly for reliable outcomes.
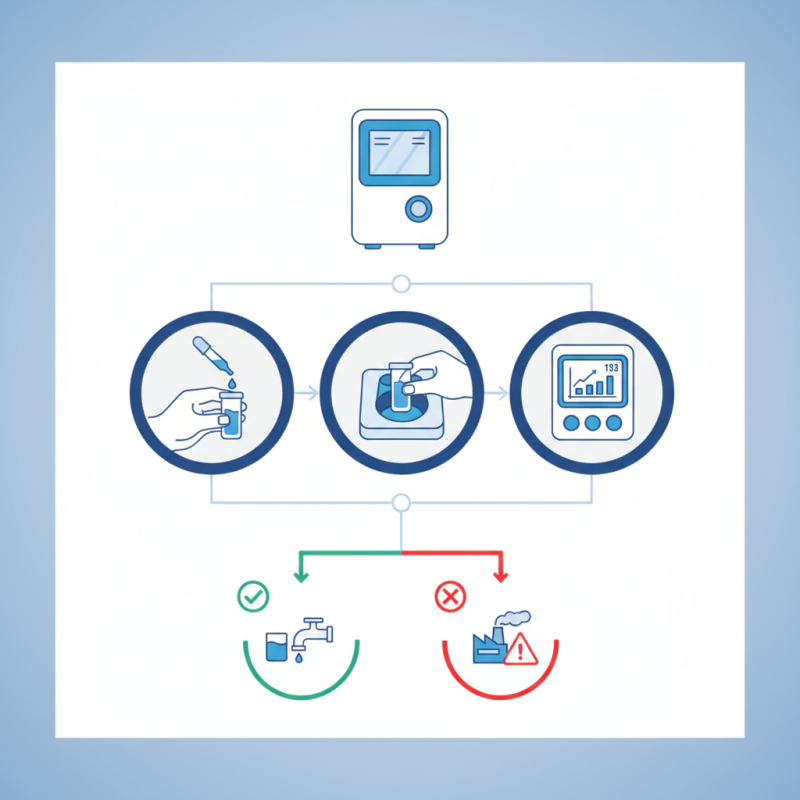
To get started, prepare the sample water carefully. The colorimeter works by comparing the color intensity of the sample to a standard. If the sample isn't clean, results can be skewed. Dull colors can confuse readings, so take the time to ensure clarity. Regular calibration is also important to maintain accuracy. The user must remember the need for routine checks, as getting an inaccurate reading can lead to flawed interpretations.
While it might seem straightforward, users often face challenges. For example, the lighting conditions can affect the readings significantly. Poor lighting may lead to misinterpretation of color. It's important to conduct tests in consistent environments. Consider documenting your process. Reflect on any discrepancies that arise during testing. Ultimately, improvement comes from learning from these experiences.

Preparing for chlorine testing with a pocket colorimeter requires careful attention to detail. Start by gathering all necessary materials. You'll need distilled water for calibration, calibration standards, and the colorimeter itself. Make sure the device is clean and fully charged. A dirty lens can lead to inaccurate readings.
Next, gather your samples. Chlorine levels can vary significantly in different sources. Collect water samples in clean, clear containers. Try to avoid bubbles while filling the containers. Bubbles can alter results and, thus, must be minimized. It’s also crucial to work in a consistent environment. Temperature and lighting may impact the readings.
Calibration of the device is another vital step. Follow the instructions carefully, ensuring you use the correct standards. Many users forget this part, leading to frustration later on. It may take a few tries to achieve accurate measurements. Don’t rush. Take your time to reflect on each step to ensure accuracy in your testing process.
Accurate chlorine testing starts with proper water sample collection. It’s not just about grabbing a sample. The quality of your water sample can impact test results significantly. Professionals recommend using clean, non-reactive containers to avoid contamination. Any residue from previous samples can skew results.
Be mindful of where you collect your samples. For instance, avoid areas with direct sunlight or heavy wave action. Research indicates that samples taken in calm conditions yield more reliable results. It’s also critical to collect samples at different depths if assessing a body of water. This method ensures a comprehensive view of chlorine levels.
Timing matters as well. Collect samples when water conditions are stable, usually early morning or late evening. Studies show that chlorine levels can fluctuate throughout the day due to varying temperatures and sunlight exposure. Taking samples during these peak times can lead to misleading data. Failing to consider these factors can result in inaccurate assessments of water safety.
To conduct a chlorine test accurately, you need to follow precise procedures. Start by preparing your sample. It’s essential to collect water samples carefully. Samples should be free from debris. Use a clean container to avoid contamination. Ideally, test the water within 24 hours of collection. The American Water Works Association recommends this for best results.
Next, calibrate your pocket colorimeter. This step is vital to ensure accuracy. According to a study published in the Journal of Environmental Monitoring, calibration can reduce testing errors by up to 15%. Select the correct wavelength for chlorine detection. Follow the device's instructions closely. Once calibrated, add a specific reagent to your sample. Wait for the recommended time before measuring. This time varies based on the reagents used.
Finally, analyze the results. Pay attention to color changes, as they indicate chlorine levels. Some tests may require visual assessment. Relying solely on automatic readings can lead to misinterpretations. It's crucial to cross-reference with standard values. If results seem inconsistent, retest your sample. This discipline in testing ensures reliable data, which is crucial for water quality management.
This bar chart represents the chlorine concentration levels in different water samples tested with the DR300 Pocket Colorimeter. The measurements are in milligrams per liter (mg/L).
Interpreting chlorine testing results with precision is crucial. The DR300 Pocket Colorimeter provides valuable insight into water quality. Understanding the significance of readings is essential. Chlorine levels directly affect water safety. The EPA suggests a chlorine concentration between 1-4 mg/L for safe drinking water.
Ensure that your colorimeter is well-calibrated for accurate results. Calibration should align with standard solutions. Deviations in calibration can mislead interpretations. A study showed that improper calibration can lead to errors exceeding 20%. Regular checks are key to maintaining accuracy.
It’s also important to consider environmental factors. Temperature can affect the reaction, skewing results. For instance, at higher temperatures, chlorine may dissipate faster. This could lead to underestimating actual levels. Be aware of these variables when interpreting your data. Attention to detail in these aspects can make a significant difference in the reliability of your testing outcomes.